FDA to add blood clot warnings on J&J coronavirus vaccines
05/13/2021 / By Ramon Tomey
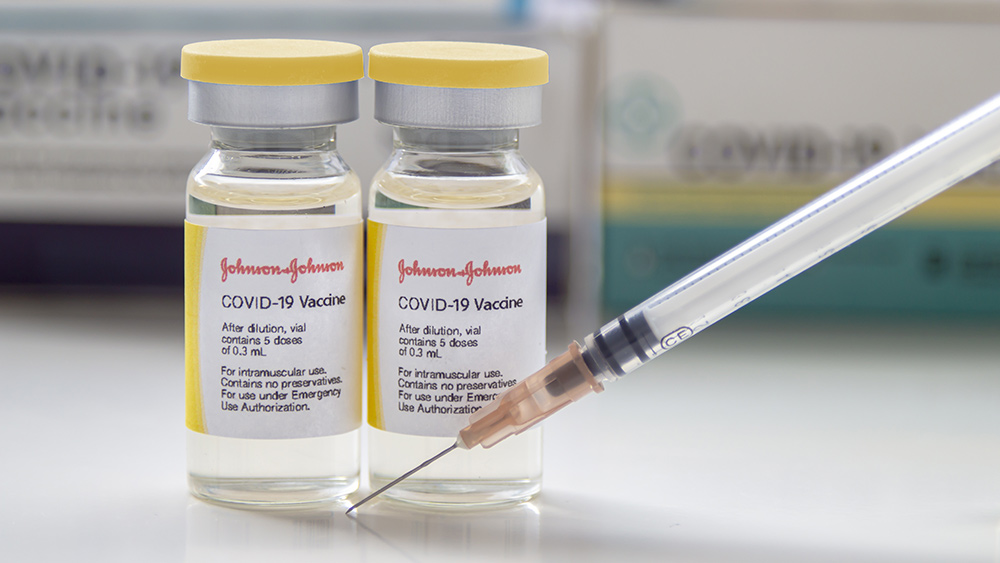
The Food and Drug Administration (FDA) said the Johnson & Johnson Wuhan coronavirus (COVID-19) vaccine will now include a warning about blood clotting risks. Its announcement came after the Centers for Disease Control and Prevention (CDC) lifted the suspension of the single-dose COVID-19 vaccine. The two government agencies previously recommended a halt on the J&J vaccine amid reports of blood clots with low platelet count in some patients.
According to the CDC’s Mortality and Morbidity Weekly Report for April 30, the FDA has added a warning for patients 18 to 49 years old regarding “clotting events … reported among vaccine recipients.” It emphasized the importance of the warning “to ensure that women aged [below] 50 years old are aware of the increased risk [of the clots] … and that other COVID-19 vaccines are available.” The report recommended that updated fact sheets for the vaccine should be provided to both recipients and their caregivers before inoculation.
The CDC’s Advisory Committee on Immunization Practices (ACIP) voted to resume use of the vaccine during its April 23 meeting. It also recommended that the FDA put a warning about the risks of blood clots, which the regulator fulfilled. The committee argued that the pause should be lifted because the benefits of the J&J vaccine outweighed its potential risks. It also said that limiting its use to specific sectors of the populations would do little to prevent hospitalizations and deaths stemming from COVID-19.
“The summary of evidence showed that the [J&J] COVID-19 vaccine is a highly effective and flexible prevention tool that can be useful in communities with increasing COVID-19 incidence and emerging variants of SARS-CoV-2. Limiting vaccine use to specific populations … could reduce number of [blood clot] cases – but could also challenge public health implementation, limit personal choice and disproportionately affect populations with barriers to vaccine access or … difficulty returning for a second [vaccine] dose,” the CDC report said.
J&J’s adenoviral vector vaccine stood out from its counterparts in a number of ways. It does not require sub-zero temperatures compared to the mRNA vaccines by Pfizer and Moderna. Furthermore, the vaccine only call for a single dose – which provided convenience. Other vaccine candidates require two doses to provide full protection. The FDA granted emergency use approval to the one-dose vaccine in late February 2021.
The CDC is also looking at the cases of blood clots and low platelet count in some inoculated Americans
FOX 5 reported that ACIP members voted 10-4 to resume use of the vaccine. CDC Director Dr. Rochelle Walensky commented on the decision: “Above all else, health and safety are at the forefront of our decisions. Our vaccine safety systems are working.” She added that the CDC has “identified exceptionally rare events” stemming from the J&J vaccine and will continue to do so. (Related: Johnson & Johnson vaccine under investigation over deadly blood clot cases.)
“We are grateful to the [ACIP] and its medical experts for the rigorous evaluation of our COVID-19 vaccine,” J&J Chief Scientific Officer Dr. Paul Stoffels remarked. He added: “As the global pandemic continues to devastate communities around the world, we believe a single-shot, easily transportable COVID-19 vaccine with demonstrated protection against multiple variants can help protect the health and safety of people everywhere.”
Concurrent with the resumption of the J&J vaccine’s use, the CDC said it is examining two new cases of blood clots. A man and a woman – both under the age of 60 – reported the clots after vaccination.
Fifteen people had already reported blood clots alongside low platelet count after getting immunized with the J&J vaccine by the time the ACIP convened its April 23 meeting. All 15 patients were hospitalized: 12 were admitted to intensive care units, while three eventually died. While 13 of the patients were women aged 18 to 49 years old, two of them were aged 50 years old and older.
Incidentally, the two 50-and-up vaccine fatalities included a woman from Oregon. Health officials in the Beaver State confirmed her death on April 22 – a day before the ACIP meeting. The CDC was informed of the case on April 18, while the Oregon Health Authority (OHA) received word of the case two days later on April 20.
According to an April 22 OHA statement, the 50-year-old Oregonian developed a “serious blood clot … in combination with very low platelets.” Both serious reactions occurred “within two weeks following vaccination,” the statement said. It added that CDC and OHA officials are looking into the matter.
The OHA statement also mentioned that the Oregonian’s death had been recorded in the CDC’s Vaccine Adverse Event Reporting System. “The case will add to the evidence of potential risk associated with [the J&J] vaccine,” it noted. However, until an investigation on the incident is complete – it cannot be concluded “whether [the woman’s] death is related to the vaccine.”
Visit VaccineInjuryNews.com to read more articles about the blood clots caused by the J&J one-dose COVID-19 vaccine.
Sources include:
Tagged Under: adverse reactions, Advisory Committee on Immunization Practices, Big Pharma, Blood clots, Centers for Disease Control and Prevention, coronavirus vaccine, covid-19 pandemic, Food and Drug Administration, Johnson & Johnson, low platelet count, Vaccine deaths, Wuhan coronavirus
RECENT NEWS & ARTICLES
COPYRIGHT © 2017 CDC NEWS